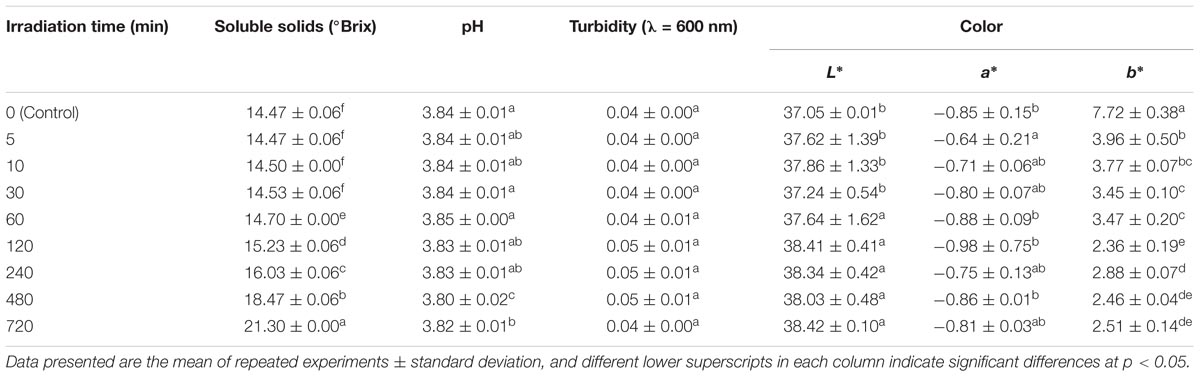
The major advantage of this technique is that nothing is added to the apple juice hence, no salt is formed at the end of the treatment.Submitted to: Journal of Agriculture and Food ChemistryĬitation: Friedman, M., Henika, P.R., Levin, C.E., Mandrell, R.E. Once the enzyme has been irreversibly inactivated, it is simply a matter of reversing the compartments and repeating the process, thereby restoring the OH − ions in the juice and reducing the acidity to its original level. As is the case when HCl is added, it is the H+ ions that have the effect of acidifying the apple juice. This technology uses electricity to literally split the water molecules present in the juice into H + and OH − ions and direct them into two compartments separated by a membrane: apple juice and the H + ions on one side, and the OH − ions on the other. The ion-selective membrane (M) prevents the juice from being diluted. Alternating compartments so that the juice returns to its initial pH (3.3 to 3.7) inhibits the enzymatic oxidation reaction in the juice caused by PPO.Circulating the juice in the acid compartment until pH=2.0, and.Sample of commercial or homemade opalescent apple juice.īipolar membrane (BPM) electrodialysis, in the presence of an electric field, generates H + and OH − ions from the hydrolysis of the water contained in the juice.Sample of PPO-oxidized, untreated opalescent apple juice.Sample of opalescent apple juice treated by means of bipolar membrane electrodialysis.The solution is bipolar membrane electrodialysis. Table salt may be good on tomatoes and cucumbers, but it lends apple juice a quite undesirable salty taste. 
However, the salt (in this case, NaCl or table salt) creates a problem. When the acid and the base interact, they cancel each other out, producing water (H 2O) and salt. However, in order to obtain a juice that does not make you pucker, the acidity must subsequently be corrected! In the laboratory, the pH can be lowered by adding an acid (such as hydrochloric acid (HCl)) and then raised by adding a base (such as sodium hydroxide or NaOH). It is therefore possible to inactivate the enzyme by acidifying the juice (down to pH 2). However, by slightly increasing the acidity of the juice, it can be inactivated: simply sprinkling lemon juice (~ pH 2) on cut apple pieces prevents browning. PPO functions very well at this level of acidity. The natural organic acids present in apple juice give it its characteristic slightly acidic taste (pH 3.3 to 3.7). Although these treatments are effective in terms of preserving colour, they do change the taste of the apple juice. The enzyme is then destroyed using a heat treatment. To remedy this problem at the industrial scale, the enzymatic browning reaction is first delayed by adding an antioxidant such as vitamin C. In a process comparable to the rusting of iron, PPO oxidizes the apple's phenolic compounds, which then clump together to form brown pigments. This browning reaction is caused by an enzyme, polyphenol oxidase (PPO). However, just like cut apples, this juice turns brown with time. Opalescent apple juice has lots of pulp, which enhances its sensory properties. This natural food product provides fibre and nutrients that may be lacking in clarified juices. Consumers are increasingly interested in opalescent or unclarified apple juice.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
